Transfected Stable Cell Lines
Reliable | High-Performance | Wide Rage
Precision reporter, kinase, immune receptor, biosimilar, Cas9, and knockout stable cell lines for diverse applications.
Cat. No. : CSC-RO0032 Host Cell : BxPC3
Size : >1x106 cells/vial Validation : T7 Endonuclease I assay
| Cat. No. | CSC-RO0032 |
| Description | BXPC-3-Cas9 cell line is engineered to stably overexpress Cas9 nuclease. The Cas9 nuclease in BXPC-3-Cas9 cell line has been functionally validated using T7 Endonuclease I assay. In combination with separately transfected sgRNAs, BXPC-3-Cas9 cell line can be used to efficiently generate targeted genomic modifications including gene knockout, gene knockin, gene mutagenesis, gene tagging etc. It is also an ideal cell line model for sgRNA screening and validation, either individually or in pools. |
| Introduction | Clustered regularly interspaced palindromic repeats (CRISPR)/Cas9 is a gene-editing technology that contains two essential components: a guide RNA (gRNA) to match a target gene, and the Cas9 (CRISPR-associated protein 9) endonuclease which causes a double-stranded DNA break, allowing modifications to the genome via nonhomologous end joining (NHEJ) or homology-directed repair (HDR). |
| Product Type | Cas9 overexpression stable cell line |
| Target Gene | Cas9 |
| Host Cell | BxPC3 |
| Host Cell Species | Homo sapiens (Human) |
| Applications |
1) CRISPR genome editing, such as gene knockout (KO), gene knockin (KI), gene mutagenesis, gene tagging etc. 2) High-throughput sgRNA screening and validation |
| Size | One vial of frozen cells, typically >1x106 cells/vial |
| Validation | T7 Endonuclease I assay |
| Quality Control |
1) T7E1 assay 2) Mycoplasma detection |
| Storage | Liquid nitrogen |
| Shipping | Dry ice |
| Revival | Rapidly thaw cells in a 37°C water bath. Transfer contents into a tube containing pre-warmed media. Centrifuge cells and seed into a 25 cm2 flask containing pre-warmed media. |
| Mycoplasma | Negative |
| Format | One frozen vial containing millions of cells |
| Storage | Liquid nitrogen |
| Safety Considerations |
The following safety precautions should be observed. 1. Use pipette aids to prevent ingestion and keep aerosols down to a minimum. 2. No eating, drinking or smoking while handling the stable line. 3. Wash hands after handling the stable line and before leaving the lab. 4. Decontaminate work surface with disinfectant or 70% ethanol before and after working with stable cells. 5. All waste should be considered hazardous. 6. Dispose of all liquid waste after each experiment and treat with bleach. |
| Ship | Dry ice |
| Target Gene | Cas9 |
Cas9, a pivotal RNA-guided DNA endonuclease, emerged as a groundbreaking discovery in 2012 within the framework of the bacterial CRISPR-Cas system. Its profound impact on molecular biology cannot be overstated. Simultaneously, BXPC-3, a human pancreatic cancer cell line, has played a crucial role in cancer research owing to its physiological relevance and genetic characteristics. The fusion of these two entities resulted in the creation of the Cas9 Stable Cell Line-BXPC-3. This milestone achievement, likely realized in the subsequent years following Cas9's discovery, marks a significant advancement in cancer biology. By integrating the Cas9 gene into the BXPC-3 genome, researchers gained a powerful tool for precise genetic manipulation within the context of pancreatic cancer.
The establishment of the Cas9 Stable Cell Line-BXPC-3 facilitates targeted genetic modifications, providing invaluable insights into the underlying mechanisms of pancreatic cancer progression and response to therapeutic interventions. This innovative cell line exemplifies the ongoing refinement and utilization of CRISPR-Cas9 technology in disease modeling and the development of targeted therapies, underscoring its transformative potential in advancing biomedical research and clinical applications.
TGF-β signaling is essential in pancreatic ductal adenocarcinoma (PDAC) development, altered in all PDAC cases as one of four major pathways. Researchers demonstrate the crucial role of SMAD2/3 in pancreatic ductal adenocarcinoma (PDAC) progression, particularly in SMAD4-null contexts. By investigating TGF-β signaling in PDAC cells lacking SMAD4, they reveal SMAD2/3's oncogenic effects, influencing collective migration through FAK and Rho/Rac signaling pathways. RNA-sequencing analyses unveil a TGF-β gene signature linked to aggressive behavior, facilitated by SMAD2/3 activation. Additionally, clinical data suggests that SMAD4-negative tumors with elevated phospho-SMAD2 levels exhibit increased aggressiveness and poorer prognosis. These findings underscore the complex interplay between TGF-β signaling components in PDAC, highlighting SMAD2/3 as potential therapeutic targets.
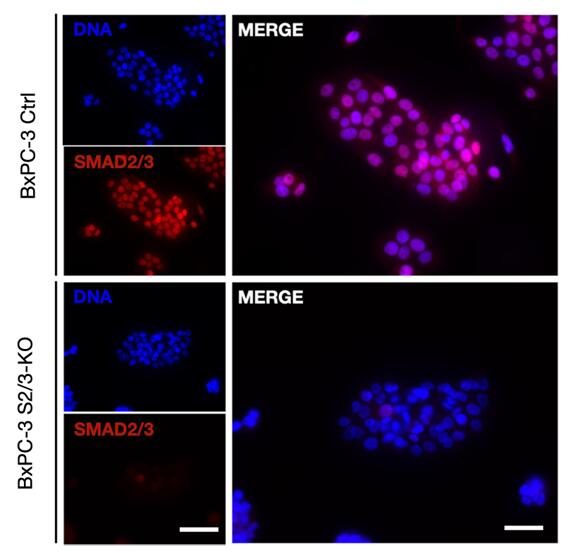 Figure 1. A double SMAD2/SMAD3 knockout BxPC-3 (SMAD4-negative) pancreatic cancer cell line was generated. SMAD protein expression and localization in response to TGF-β treatment were assessed. Altered SMAD2/3 levels and distribution were observed in knockout cells compared to controls, providing insights into TGF-β signaling in pancreatic cancer progression. (Bertrand-Chapel A, et al., 2022)
Figure 1. A double SMAD2/SMAD3 knockout BxPC-3 (SMAD4-negative) pancreatic cancer cell line was generated. SMAD protein expression and localization in response to TGF-β treatment were assessed. Altered SMAD2/3 levels and distribution were observed in knockout cells compared to controls, providing insights into TGF-β signaling in pancreatic cancer progression. (Bertrand-Chapel A, et al., 2022)
Utilizing Creative Biogene's Cas9 Stable Cell Line-BXPC-3 streamlines the process of SMAD2/SMAD3 gene knockout experiments. This stable cell line is directly amenable to CRISPR/Cas9 gene editing technology, facilitating the rapid generation of SMAD2/SMAD3 knockout cell lines, thereby saving time and resources. Such cell lines can be instrumental in investigating the TGF-β signaling pathway and its implications in pancreatic cancer progression, enhancing the convenience and efficiency of related disease research endeavors.
1. Gene Editing: Utilizing Cas9 Stable Cell Line-BXPC-3 for targeted gene knockout studies in pancreatic cancer research.
2. Drug Screening: Employing Cas9 Stable Cell Line-BXPC-3 to assess drug efficacy and resistance mechanisms in pancreatic cancer cells.
3. Functional Genomics: Investigating the role of specific genes in pancreatic cancer progression using Cas9 Stable Cell Line-BXPC-3.
4. Therapeutic Development: Validating potential therapeutic targets through CRISPR/Cas9-mediated gene editing in BXPC-3 cells.
5. Metastasis Study: Examining the impact of gene mutations on pancreatic cancer metastasis using Cas9 Stable Cell Line-BXPC-3.
6. Immunotherapy Evaluation: Assessing immune checkpoint inhibitors' effectiveness in BXPC-3 cells modified with Cas9 technology.
7. Tumor Microenvironment Research: Exploring interactions between pancreatic cancer cells and their microenvironment using Cas9 Stable Cell Line-BXPC-3.
8. Cancer Stem Cell Investigation: Characterizing cancer stem cells in pancreatic cancer using Cas9 Stable Cell Line-BXPC-3.
A: THP-1 cells were likely selected for their monocytic lineage and expression of CD19, making them suitable for evaluating the functionality of the Anti-hCD19-CAR(3G) construct in targeting CD19-positive cells.
A: Stability and expression were likely assessed through flow cytometry, immunoblotting, or functional assays measuring CAR-mediated cytotoxicity or cytokine release, with appropriate selection pressure applied.
A: Characterization may involve analysis of CAR expression levels, binding affinity to CD19, activation-induced signaling pathways, and functional implications in antigen-specific killing of CD19-positive cells.
A: Quality control likely included confirmation of CAR expression and functionality, assessment of off-target effects, and validation of phenotypic changes associated with CAR activation.
A: Comparative analysis with primary T cells or in vivo models helps validate the efficacy and safety of Anti-hCD19-CAR(3G) therapy, guiding its development as a promising treatment for B cell malignancies such as leukemia and lymphoma.
If your question is not addressed through these resources, you can fill out the online form below and we will answer your question as soon as possible.
Remarkable reliability! The Cas9 Stable Cell Line in BXPC-3 cells has been indispensable in our genome editing studies. Its stable expression of Cas9 has facilitated precise and efficient gene modifications, enabling us to dissect the functional significance of specific genetic alterations in cancer.
Trustworthy and effective! The stable expression of Cas9 in BXPC-3 cells has allowed us to perform targeted genome editing with high accuracy and fidelity. Its consistent Cas9 expression has ensured reliable and reproducible results, enhancing the reliability of our experiments.
Streamlining our research! With the Cas9 Stable Cell Line, we've been able to investigate the role of specific genes in cancer development and progression with confidence. Its stable expression of Cas9 has streamlined experimental workflows and accelerated the pace of our research.
Impressive performance! The Cas9 Stable Cell Line has consistently exhibited efficient Cas9-mediated genome editing, exceeding our expectations. Its stable expression has been instrumental in unraveling the genetic determinants of cancer phenotypes and identifying potential therapeutic targets.
A valuable tool in cancer genomics! The Cas9 Stable Cell Line has transformed our understanding of cancer biology. Its stable expression of Cas9 has provided a versatile platform for exploring gene function and dissecting molecular mechanisms underlying tumorigenesis.
Write a review of your use of Biogene products and services in your research. Your review can help your fellow researchers make informed purchasing decisions.