• Adenovirus Service • AAV Service • Lentivirus Service • Retrovirus Service


Melanoma is the most dangerous type of human skin cancer, especially NRAS mutant melanoma, which has become a difficult problem in the medical community due to its extremely high invasiveness and poor prognosis. At present, this cancer is mostly indifferent to existing immunotherapy and targeted drugs, leaving patients and doctors helpless.
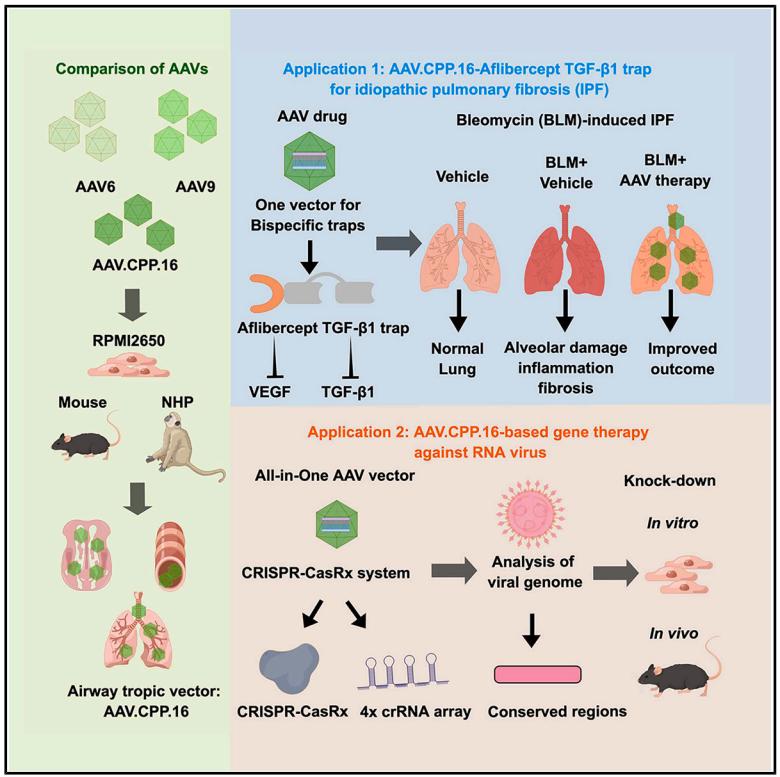
Recently, researchers published a research paper titled "Cross-species tropism of AAV.CPP.16 in the respiratory tract and its gene therapies against pulmonary fibrosis and viral infection" in Cell Reports Medicine, a subsidiary of Cell. The study showed that AAV.CPP.16 has a high tropism for respiratory tissues across species (including mice and non-human primates), and verified the gene supplementation and gene editing therapy delivered by nasal administration of AAV.CPP.16 for the treatment of idiopathic pulmonary fibrosis and viral infection. This study shows that AAV.CPP.16 is a promising vector for gene therapy and gene editing in the respiratory system and lungs.
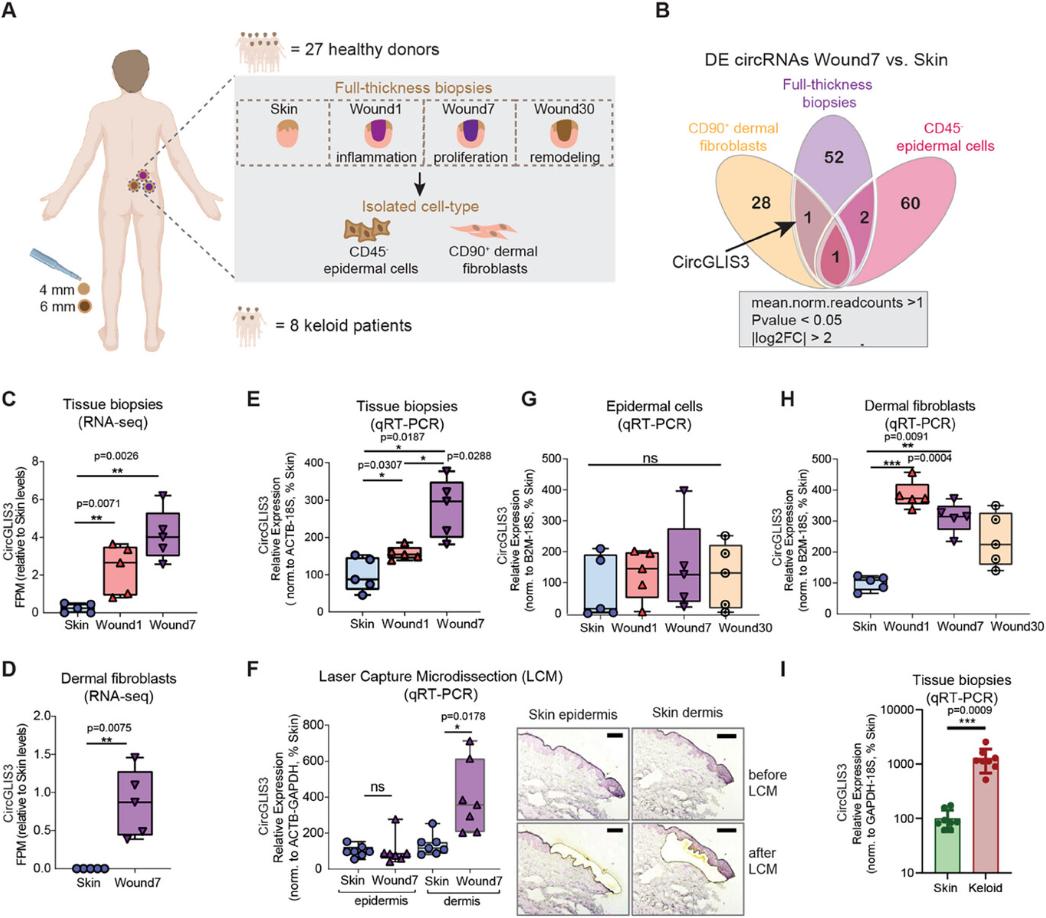
In the frontier field of medical research, skin wound healing has always been the focus of scientists. Whether it is accidental trauma, surgical incision, or skin ulcer caused by chronic disease, the speed and quality of wound healing directly affect the patient's recovery process. However, there are many complex molecular mechanisms in the skin repair process, some of which have not yet been fully revealed.
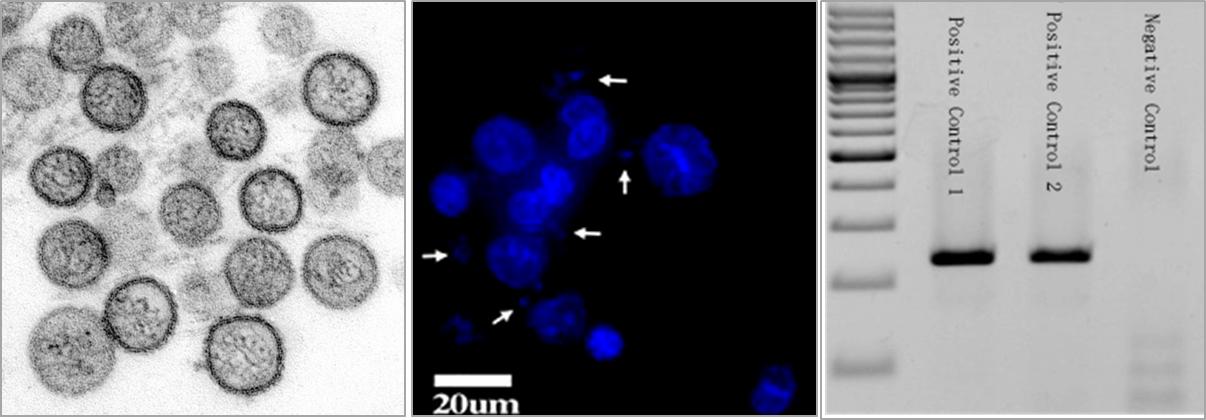
What is mycoplasma?
Low transfection efficiency

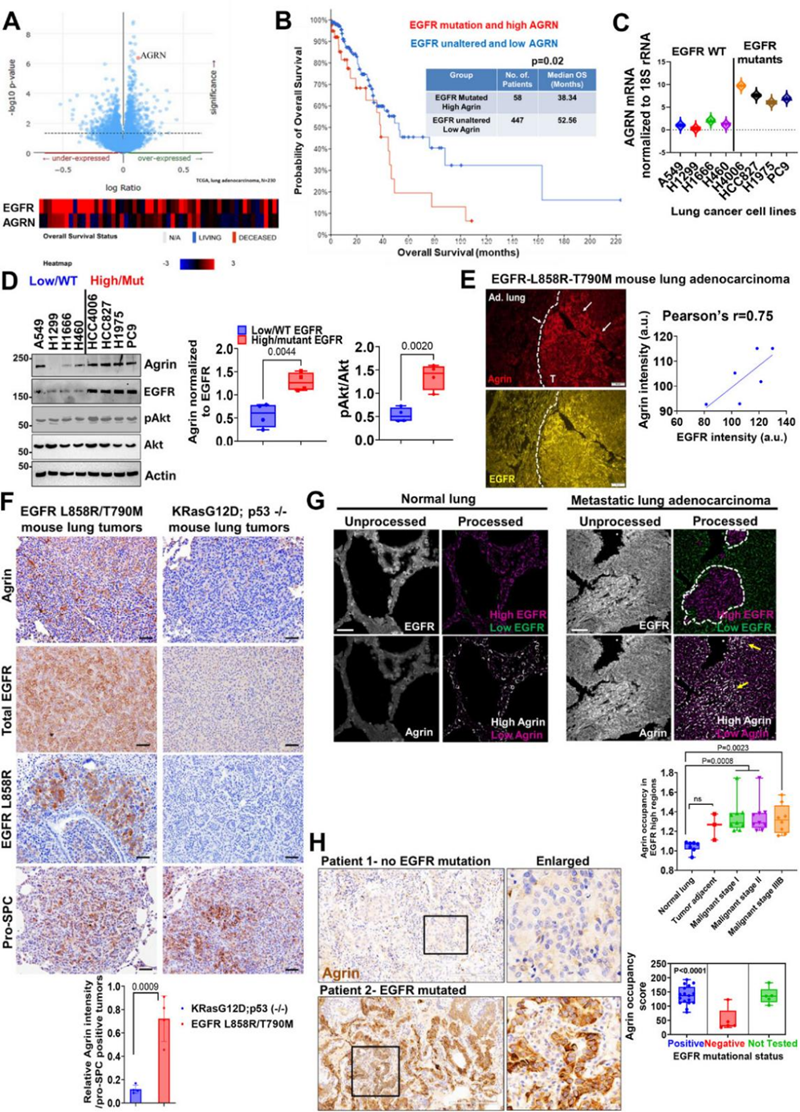
Today, lung cancer is one of the leading causes of cancer-related deaths worldwide, especially lung adenocarcinoma (LUAD) in non-small cell lung cancer, which accounts for the majority of lung cancer cases. Although targeted therapy and immunotherapy have made significant progress in recent years, the high recurrence rate and drug resistance of lung cancer remain a huge challenge in clinical practice. Fortunately, scientists have never stopped exploring.
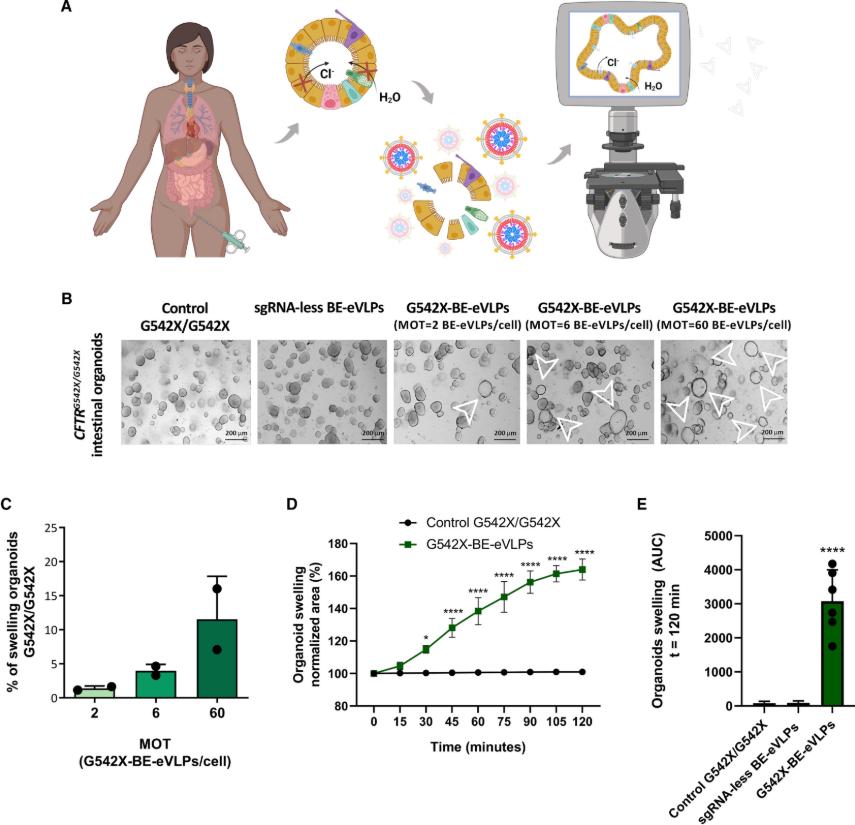
Around the world, about 162,000 people are struggling with cystic fibrosis (CF), a serious genetic disease. CF is an autosomal recessive genetic disease that severely shortens the lifespan of patients. It is caused by loss-of-function mutations in the CF transmembrane conductance regulator (CFTR) gene. These mutations affect the synthesis, folding, transport, and gating properties of the CFTR anion channel, thereby interfering with the transport of chloride and bicarbonate ions. Although modulatory drugs have made some progress in the treatment of CF, existing modulatory drugs are powerless for patients carrying specific mutations, such as those with the G542X mutation. Recently, a study published in iScience titled "Adenine base editing with engineered virus-like particles rescues the CFTR mutation G542X in patient-derived intestinal organoids" has brought new hope.
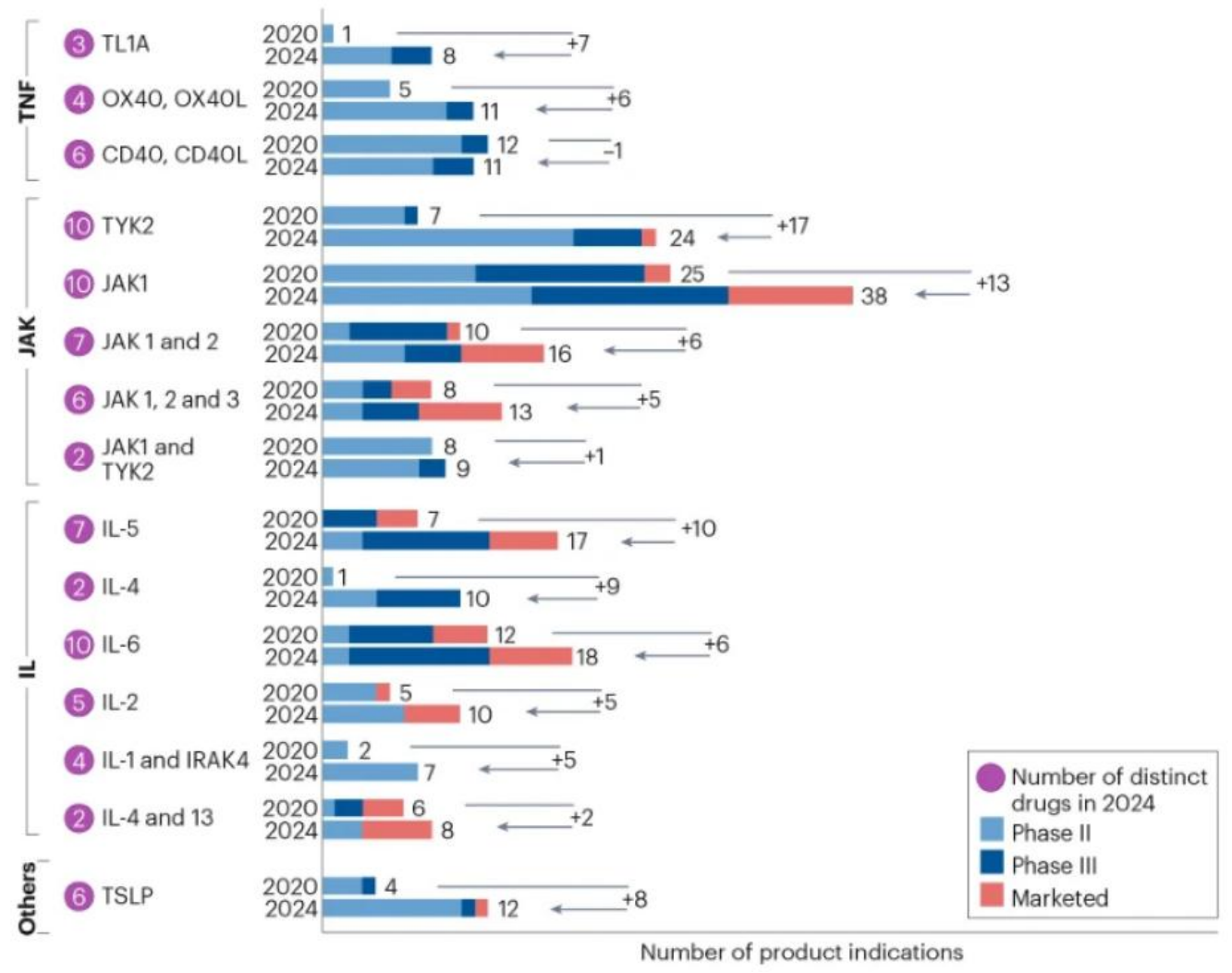
Autoimmune diseases are the result of the immune system "killing each other", and the core mechanism is the disorder of the immune system's self-recognition function, which leads to inflammatory response and tissue damage. There are more than 100 known autoimmune diseases, including systemic lupus erythematosus, rheumatoid arthritis, psoriasis, multiple sclerosis, type 1 diabetes, etc.
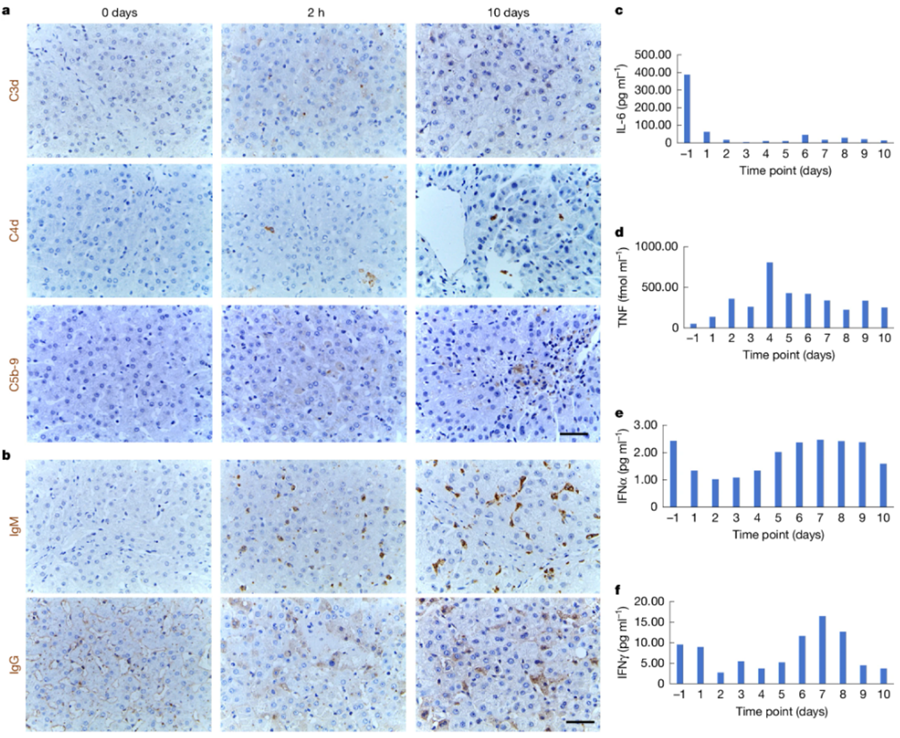
Recently, the U.S. Food and Drug Administration (FDA) officially approved a groundbreaking medical trial - using gene-edited pig livers to treat patients with liver failure! Patients with severe liver failure who cannot receive human organ transplants are temporarily connected to pig livers outside the body, which filter their blood. This is a huge breakthrough in the field of xenotransplantation, and it also brings new hope of survival to countless patients waiting for organ transplants.
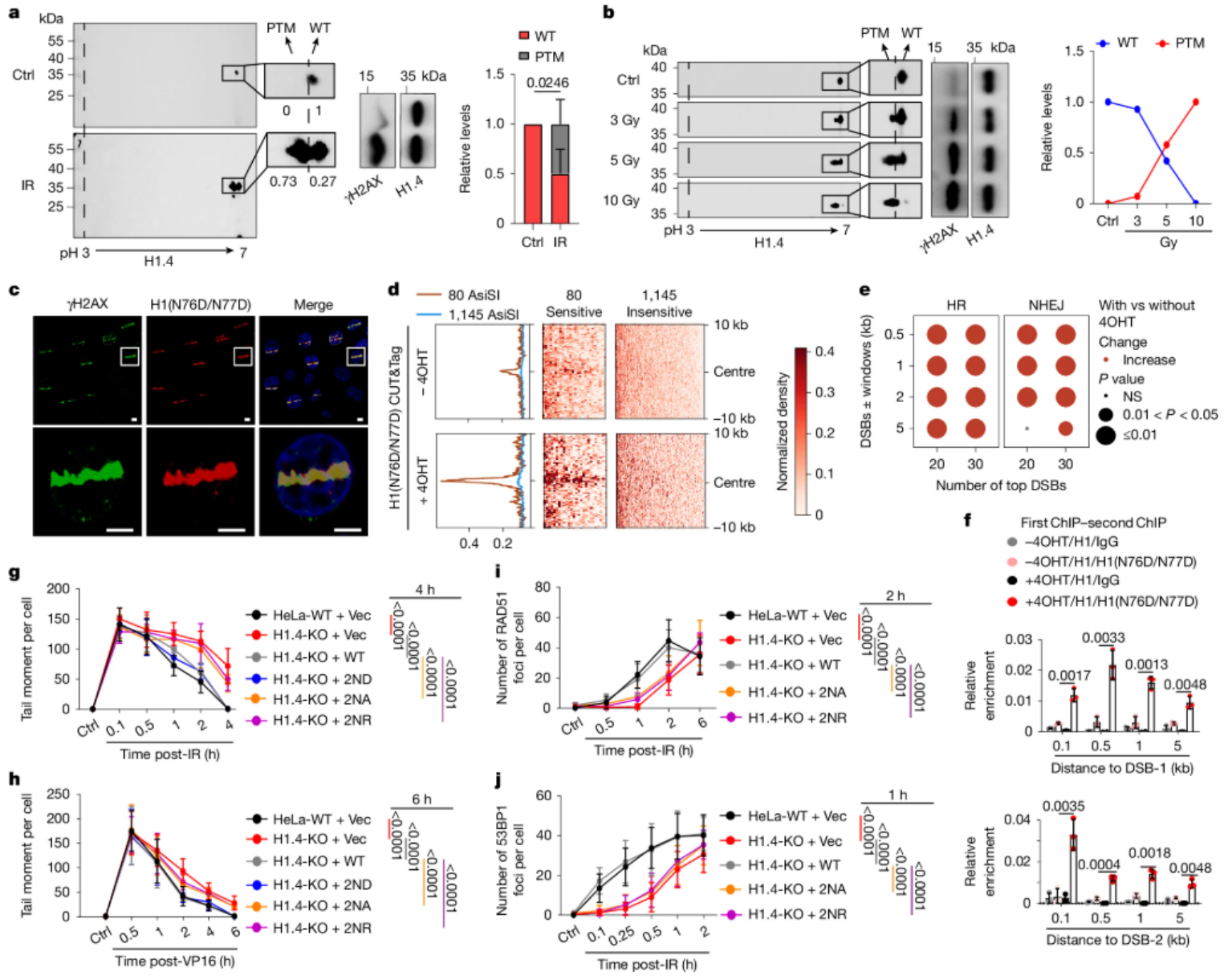
Cancer treatment has always been the focus of scientists around the world. From traditional surgery and radiotherapy to today's targeted therapy and immunotherapy, every technological advancement has brought new hope to patients. However, the complexity of cancer requires us to continue to explore the molecular mechanisms behind it.