• Adenovirus Service • AAV Service • Lentivirus Service • Retrovirus Service


Hyperuricemia (HU) is a metabolic disease caused by high serum uric acid (SUA) levels due to insufficient renal excretion, excessive production, or insufficient intestinal excretion. Various conventional treatments are commonly used to treat HU, such as valproic acid and allopurinol (xanthine oxidase inhibitor). Valproic acid increases urination, thereby enhancing the excretion of urate crystals, while allopurinol relieves symptoms by reducing uric acid production by inhibiting xanthine oxidase. However, increased urination increases the excretion of urate crystals, leading to kidney damage.
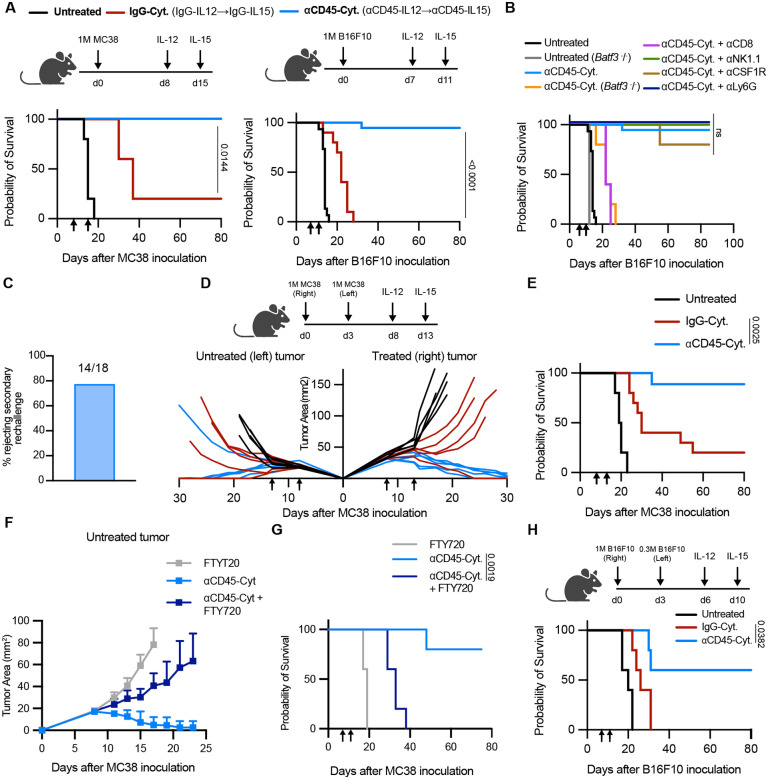
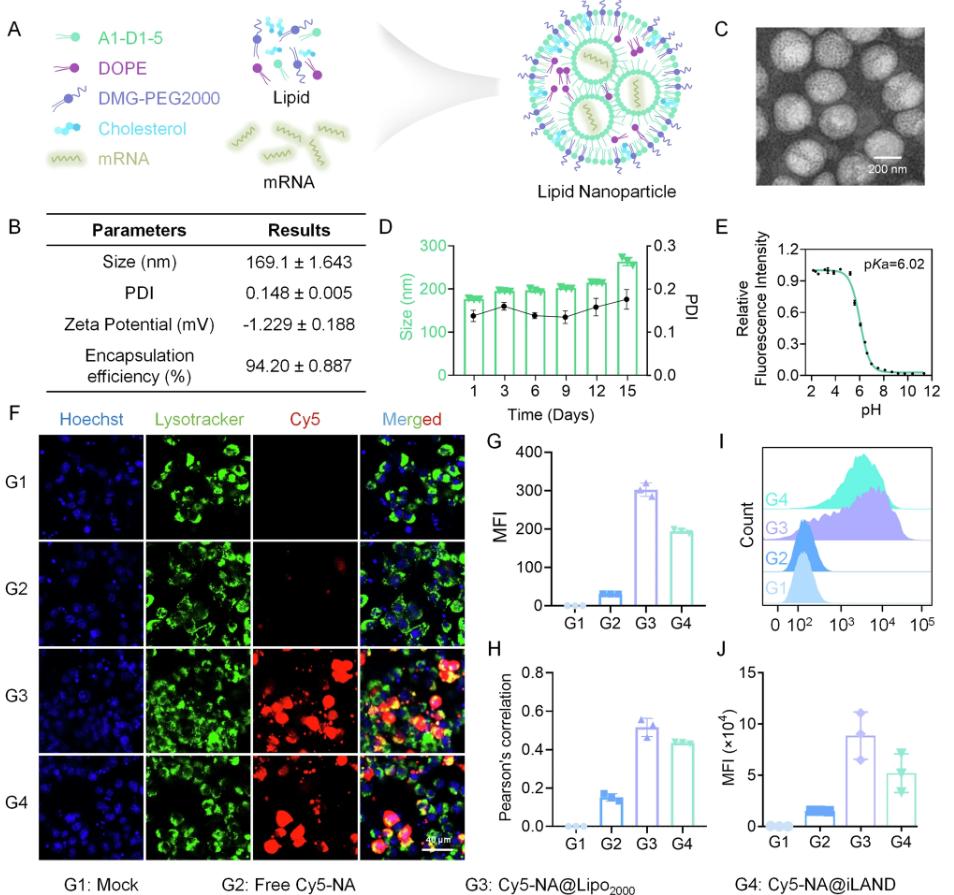
Cytokines are a class of small proteins that play key regulatory roles in immune signaling cascades. Due to their multifunctional roles in lymphocytes, cytokines have long been considered promising cancer immunotherapeutics. Systemically administered cytokines are potent immunotherapeutics but can cause severe dose-limiting toxicities.
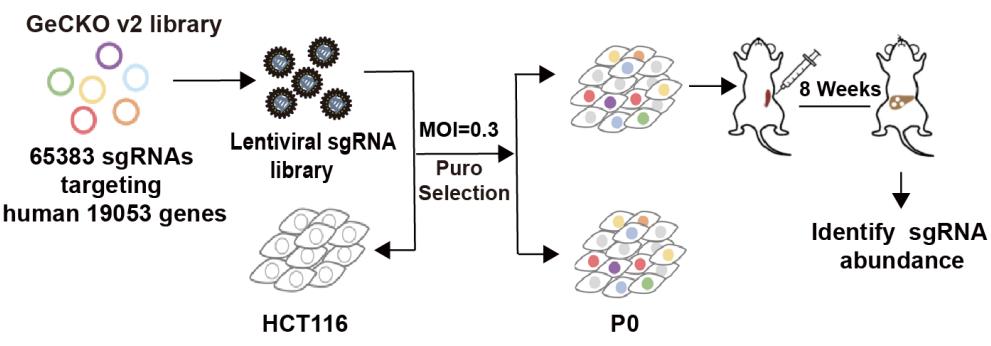
The selective metastasis of cancer cells to specific organs is a complex process that is influenced not only by anatomical factors but also by biological and organ-specific microenvironmental factors. The pre-metastatic niche refers to soluble factors and extracellular vesicles (EVs) produced by cells at the primary tumor site, which can modify the microenvironment of distant organs to accommodate migrating cancer cells and promote their growth. In addition, disseminated tumor cells (DTCs) can enter a dormant state in the circulation and in the new tissue environment, evade immune surveillance, and interact with the tissue microenvironment to awaken from dormancy. Finally, metastatic colonization requires multiple biological processes. These processes rely on the intrinsic properties of cancer cells and the permissive tumor microenvironment provided by cells in the target organ.
On August 14, 2024, the World Health Organization (WHO) announced that the monkeypox (mpox) outbreak in parts of Africa constitutes a "Public Health Emergency of International Concern" (PHEIC). This is the second time in two years that the WHO has declared a monkeypox outbreak a PHEIC event. PHEIC is the highest alert level under international health law. The WHO said that the monkeypox outbreak has the potential to spread further in Africa and to other continents. According to a report released by the Africa Centers for Disease Control and Prevention, from the beginning of this year to July 28, the number of new monkeypox cases in Africa has surged by 160% compared with the same period last year. So far, 34 African countries have reported the discovery of the disease or have been identified as "high-risk" countries.
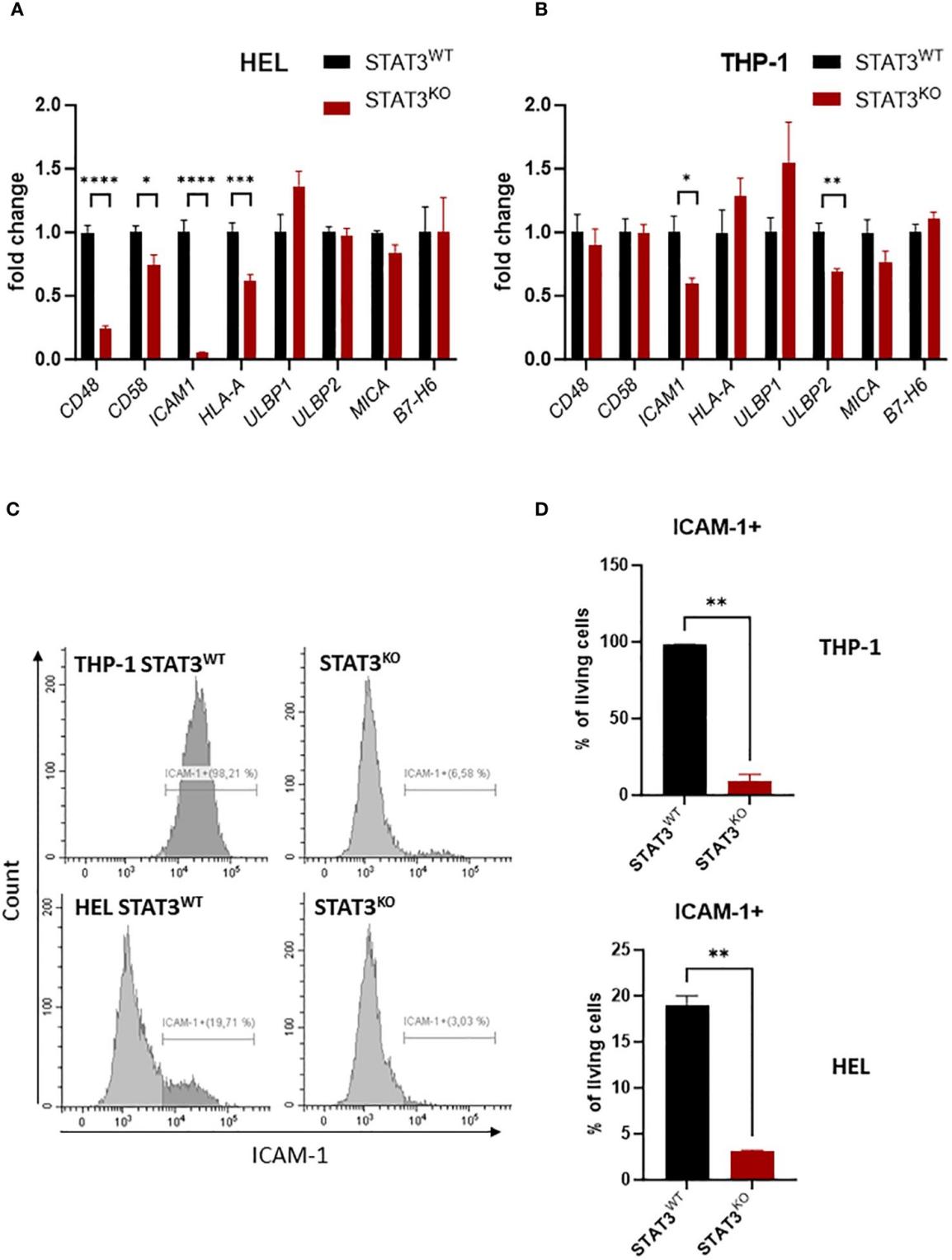
Acute myeloid leukemia (AML) is a heterogeneous disease characterized by clonal expansion of myeloid progenitor cells. Recently, in a research report entitled "STAT3 in acute myeloid leukemia facilitates natural killer cell-mediated surveillance" published in the international journal Frontiers in Immunology, scientists from Karl-Landsteiner University of Health Sciences in Austria and other institutions found that STAT3 protein may help the immune system identify leukemia cells. The relevant research results are very important for the development of new immunotherapies in the future.
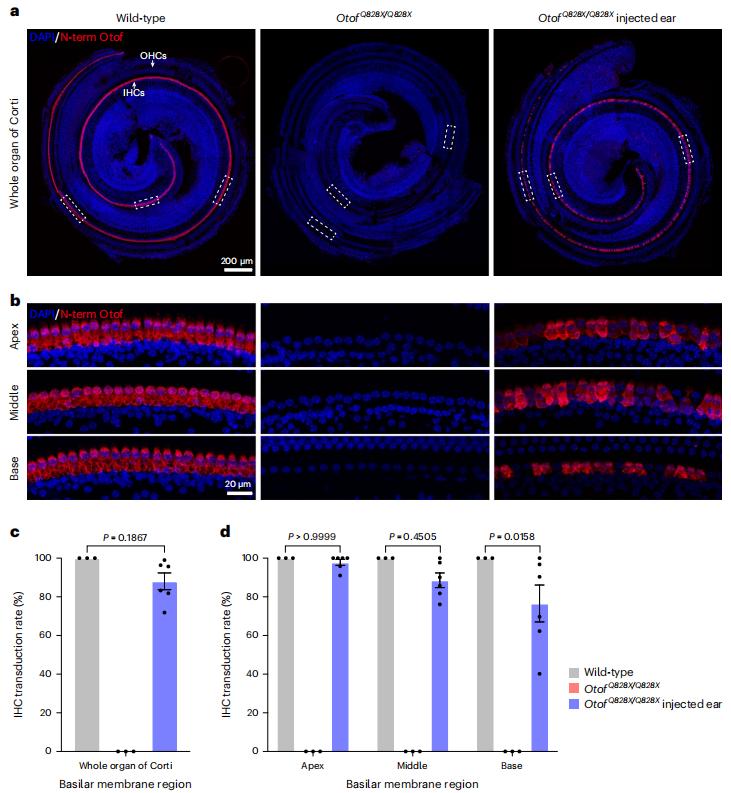
Recently, the team led by Professor Yilai Shu from the Affiliated Eye, Ear, Nose and Throat Hospital of Fudan University in China published a research paper titled "A base editor for the long-term restoration of auditory function in mice with recessive profound deafness" in the journal Nature Biomedical Engineering. The study used adenine base editor (ABE)-mediated gene editing therapy to effectively repair the Otof pathogenic mutation in the deaf mouse model and restore the expression level of otoferlin in 88% of the inner hair cells of the inner ear. At the same time, it improved the synaptic exocytosis function of the inner hair cells of the inner ear and restored hearing to a level close to the wild type for up to 1.5 years without obvious off-target effects. It is reported that this is the longest-observed effective result in the field of gene therapy for deafness in animal models to date.
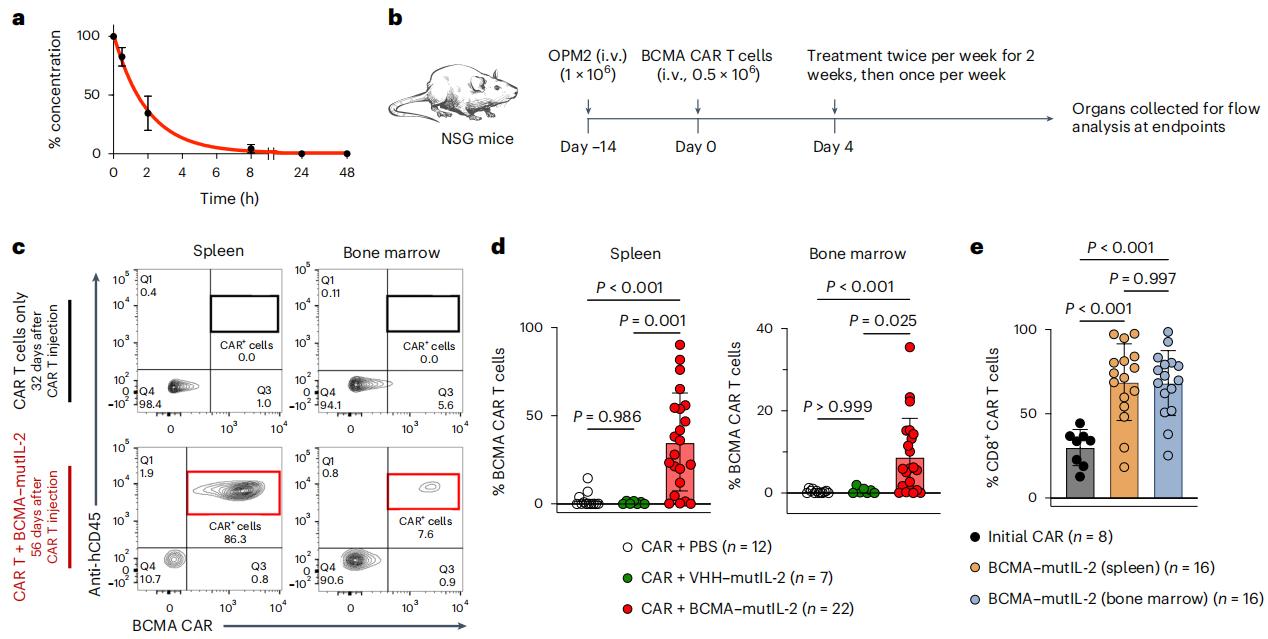
Although CAR-T cell therapy has brought revolutionary progress to the field of cancer treatment, especially in the treatment of certain B-cell blood cancers such as leukemia, lymphoma and multiple myeloma, this therapy still faces a major challenge: many patients will eventually relapse even after a short period of complete remission.
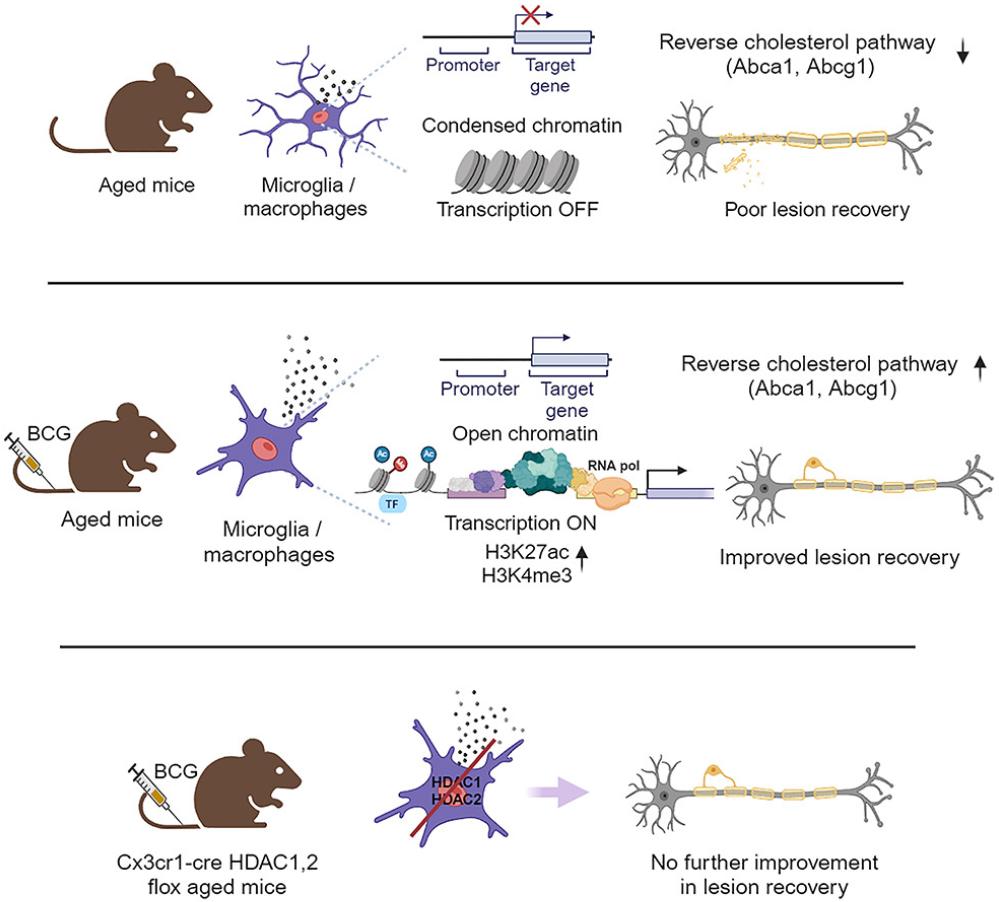
Recently, researchers from the Technical University of Munich in Germany published a research paper titled "Innate immune training restores pro-reparative myeloid functions to promote remyelination in the aged central nervous system" in the Cell journal Immunity. The study showed that age-related epigenomic changes impair the regenerative function of microglia, but innate immune training can reverse these changes and enhance their pro-regenerative ability in demyelinating injuries. These findings provide new insights into the aging-related decline of myeloid function and how to prevent this decline through innate immune reprogramming.
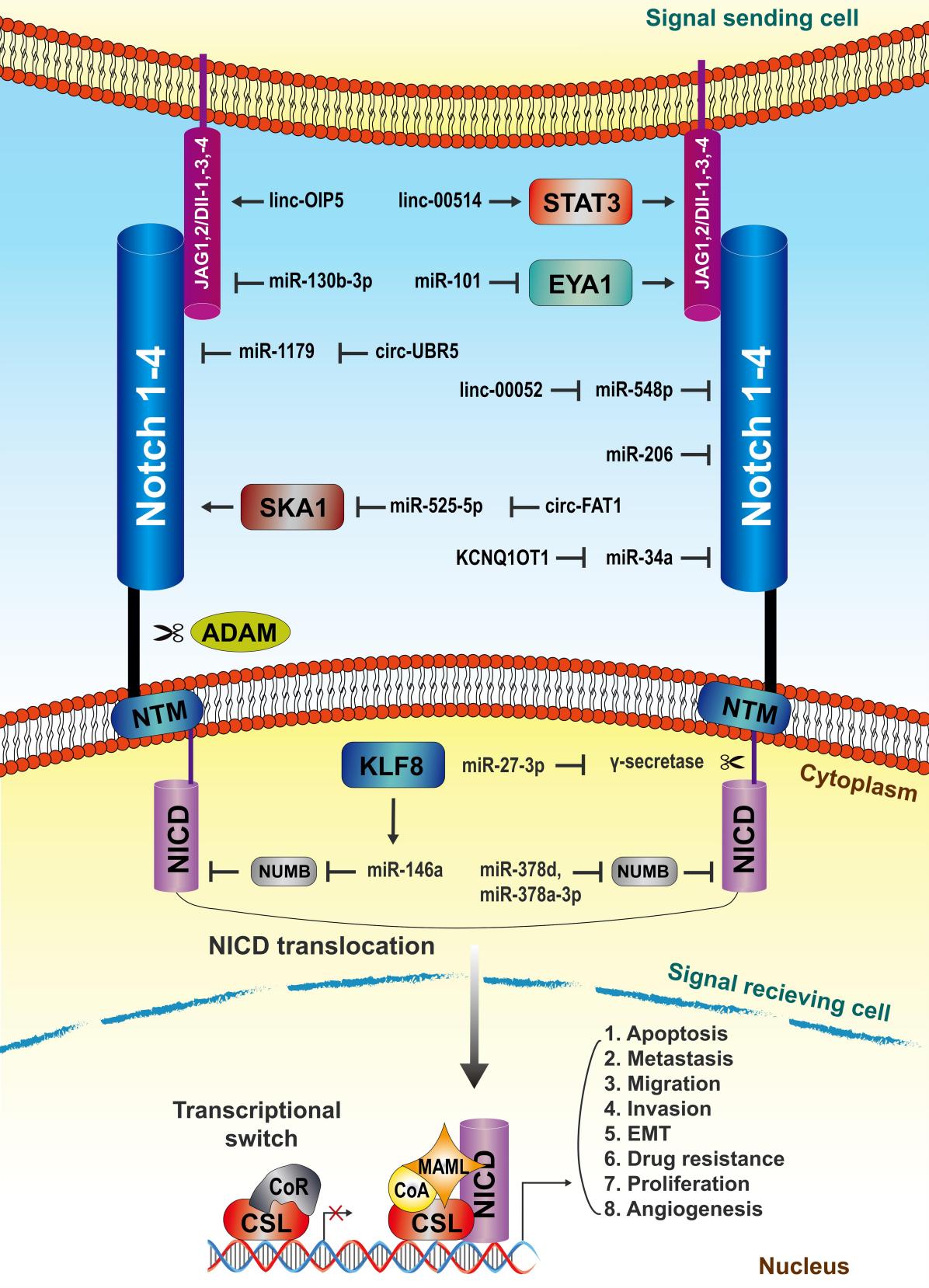
Breast cancer remains one of the most challenging cancers, mainly due to its heterogeneity and metastatic propensity. The Notch signaling pathway is required for multiple cellular processes and is involved in the development and progression of breast cancer. Noncoding RNAs (ncRNAs) have now become key regulators of gene expression, which can affect the biology of cancer by interacting with the Notch signaling pathway. Noncoding RNAs include microRNAs, long noncoding RNAs (lncRNAs), and circular RNAs (circRNAs). Recently, in a review report published in the international journal Gene Expression, entitled "Non-coding RNAs Affect Breast Cancer Development Through the Notch Signaling Pathway: An Overview", researchers from Tabriz University outlined the importance of ncRNAs in human breast cancer and their potential as therapeutic targets, paving the way for the development of innovative therapies to significantly impact patient care and prognosis.
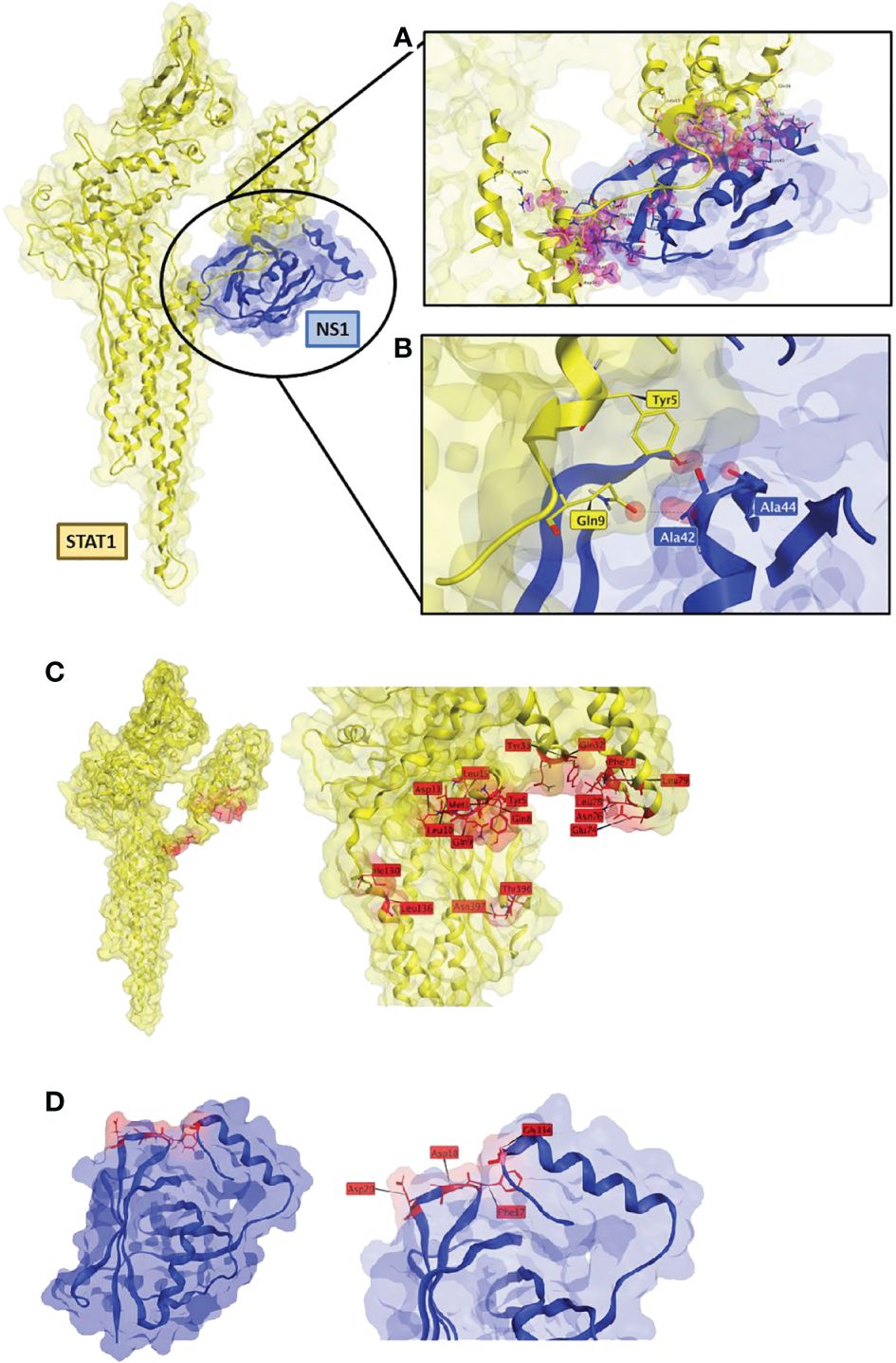
Respiratory syncytial virus (RSV) causes a significant disease burden in the global population, with an estimated 33.1 million cases each year, and is the leading cause of bronchitis and viral pneumonia in infants. It particularly affects children and the elderly, and treatment options are limited and relatively ineffective.